Case Studies In Modern Drug Discovery And Development: A Comprehensive Guide To The Process

Drug discovery and development is a complex and challenging process. It can take many years and billions of dollars to bring a new drug to market. The book Case Studies In Modern Drug Discovery And Development provides a comprehensive guide to the process, from target identification to clinical trials. The book is written by a team of experts in the field and is essential reading for anyone involved in drug discovery and development.
5 out of 5
| Language | : | English |
| File size | : | 21653 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 980 pages |
| Lending | : | Enabled |
Target Identification
The first step in drug discovery is target identification. This involves identifying a specific molecule or pathway that is involved in a disease process. Once a target has been identified, researchers can begin to develop drugs that target that molecule or pathway.
Preclinical Development
Once a target has been identified, researchers begin preclinical development. This involves testing the drug in animal models to assess its safety and efficacy. Preclinical development also includes studies to determine the drug's pharmacokinetics and pharmacodynamics.
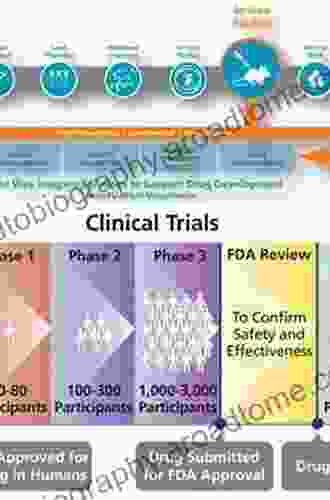
Clinical Trials
Once a drug has been shown to be safe and effective in animal models, it can enter clinical trials. Clinical trials are studies that are conducted in humans to assess the safety and efficacy of a drug. Clinical trials are typically divided into three phases:
- Phase I trials: These trials are small studies that are designed to assess the safety of a drug.
- Phase II trials: These trials are larger studies that are designed to assess the efficacy of a drug.
- Phase III trials: These trials are large studies that are designed to confirm the safety and efficacy of a drug.
Regulatory Approval
Once a drug has been shown to be safe and effective in clinical trials, it must be approved by a regulatory agency before it can be marketed. The regulatory approval process can be complex and time-consuming. In the United States, the Food and Drug Administration (FDA) is responsible for approving new drugs.
Marketing
Once a drug has been approved by a regulatory agency, it can be marketed to patients. Marketing a drug involves creating awareness of the drug and educating patients about its benefits. Drug marketing can be a complex and challenging process.
Case Studies
The book Case Studies In Modern Drug Discovery And Development includes a number of case studies that illustrate the drug discovery and development process. These case studies provide real-world examples of how drugs have been brought to market. The case studies are a valuable resource for anyone involved in drug discovery and development.
Case Studies In Modern Drug Discovery And Development is a comprehensive guide to the drug discovery and development process. The book is written by a team of experts in the field and is essential reading for anyone involved in drug discovery and development.
5 out of 5
| Language | : | English |
| File size | : | 21653 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 980 pages |
| Lending | : | Enabled |
Do you want to contribute by writing guest posts on this blog?
Please contact us and send us a resume of previous articles that you have written.
 Book
Book Novel
Novel Page
Page Chapter
Chapter Text
Text Story
Story Genre
Genre Reader
Reader Library
Library Paperback
Paperback E-book
E-book Magazine
Magazine Newspaper
Newspaper Paragraph
Paragraph Sentence
Sentence Bookmark
Bookmark Shelf
Shelf Glossary
Glossary Bibliography
Bibliography Foreword
Foreword Preface
Preface Synopsis
Synopsis Annotation
Annotation Footnote
Footnote Manuscript
Manuscript Scroll
Scroll Codex
Codex Tome
Tome Bestseller
Bestseller Classics
Classics Library card
Library card Narrative
Narrative Biography
Biography Autobiography
Autobiography Memoir
Memoir Reference
Reference Encyclopedia
Encyclopedia Archibald D Hart
Archibald D Hart Abraham Myerson
Abraham Myerson Wayne Gladstone
Wayne Gladstone Jawanza Kunjufu
Jawanza Kunjufu Stephen Fulder
Stephen Fulder Lee Wilson
Lee Wilson Peter Paravalos
Peter Paravalos Mary Beth Augustine
Mary Beth Augustine 2002nd Edition Kindle Edition
2002nd Edition Kindle Edition David Lintonbon Do
David Lintonbon Do Will James
Will James Wendell Potter
Wendell Potter Linda Carol Jones
Linda Carol Jones Glenn Nystrup M S
Glenn Nystrup M S Terry E Miller
Terry E Miller Ken Warren
Ken Warren Mary Fisher Day
Mary Fisher Day James Lake
James Lake Joshua B Edel
Joshua B Edel Brian Thorne
Brian Thorne
Light bulbAdvertise smarter! Our strategic ad space ensures maximum exposure. Reserve your spot today!

 Eddie PowellUnveiling Innovations in Governmental Accounting: A Comprehensive Guide to...
Eddie PowellUnveiling Innovations in Governmental Accounting: A Comprehensive Guide to... Robert HeinleinFollow ·19.3k
Robert HeinleinFollow ·19.3k Mario BenedettiFollow ·10.4k
Mario BenedettiFollow ·10.4k Braeden HayesFollow ·5.7k
Braeden HayesFollow ·5.7k Richard AdamsFollow ·10.4k
Richard AdamsFollow ·10.4k Ralph TurnerFollow ·16.7k
Ralph TurnerFollow ·16.7k Harry CookFollow ·11.2k
Harry CookFollow ·11.2k Andy HayesFollow ·4.8k
Andy HayesFollow ·4.8k Hugh BellFollow ·11.6k
Hugh BellFollow ·11.6k

 Nathan Reed
Nathan ReedProgress In Complex Systems Optimization Operations...
This book presents...

 Duncan Cox
Duncan CoxHSK Chinese Grammar: The Ultimate Guide to Master Chinese...
HSK Chinese...

 Owen Simmons
Owen SimmonsDevelopment and Applications in Policy Support...
Unveiling the Transformative...

 Travis Foster
Travis FosterTransform Emotions Into Energy To Achieve Your Greatest...
Do you feel like your...

 Joe Simmons
Joe SimmonsUnlocking the Frontiers of Artificial Intelligence: Delve...
In the annals of artificial...
5 out of 5
| Language | : | English |
| File size | : | 21653 KB |
| Text-to-Speech | : | Enabled |
| Screen Reader | : | Supported |
| Enhanced typesetting | : | Enabled |
| Print length | : | 980 pages |
| Lending | : | Enabled |